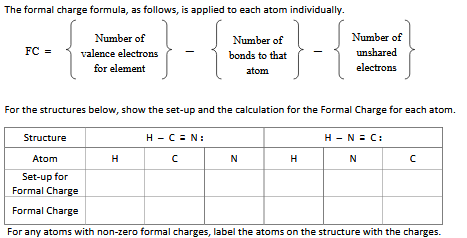
In the first case carbon and oxygen both have a charge of zero, resulting in zero overall charges. Notice that in the case of carbon dioxide, every possible formation of the molecule has a formal charge of zero. Photo: By Bkwan740 at English Wikipedia – Transferred from en.wikipedia to Commons by Ronhjones using CommonsHelper., Public Domain, 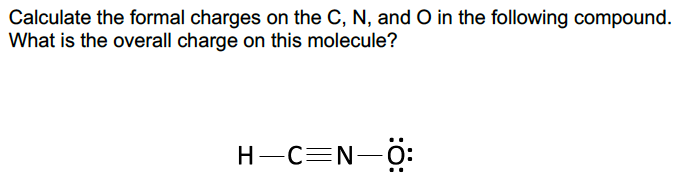
Finally, the carbon atom could be joined to both oxygen atoms with single bonds.Įxample of calculations for formal charge of carbon dioxide. The carbon atom could potentially be joined to the oxygen atoms surrounding it with double bonds, or the carbon atom could be joined to one of the oxygen atoms with a double bond and to the other oxygen atom with a single bond. Drawing the Lewis structure of the molecule reveals that it can be sketched out in three different ways. Let’s look at an example of formal charge calculation:Ĭarbon dioxide, CO2, is a neutral molecule that possesses 16 electrons in its valence shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
